Our Therapeutic Focus
Tourette Syndrome
Tourette syndrome (TS) is a neurological disorder characterized by motor and vocal tics that begin in childhood and persist over time. The tics are brief, spontaneous and irregular.
It is estimated that 1 out of every 160 children (ages 5-17) in the U.S. has TS, with symptoms continuing into adulthood for many patients.
Restless Legs Syndrome with Augmentation
This condition is an extension of restless legs syndrome (RLS), also known as Willis-Ekbom Disease, a neurological sensory disorder characterized by an irresistible urge to move one’s legs due to uncomfortable sensations. Restless legs syndrome with augmentation (RLSa) is the worsening of RLS symptoms despite treatment with conventional therapies.
According to the International Parkinson and Movement Disorder Society, approximately 5%-10% of the general population is impacted by RLS, making it the most common movement disorder.
Clinical Trials
Phase 3 Clinical Trial for Tourette Syndrome
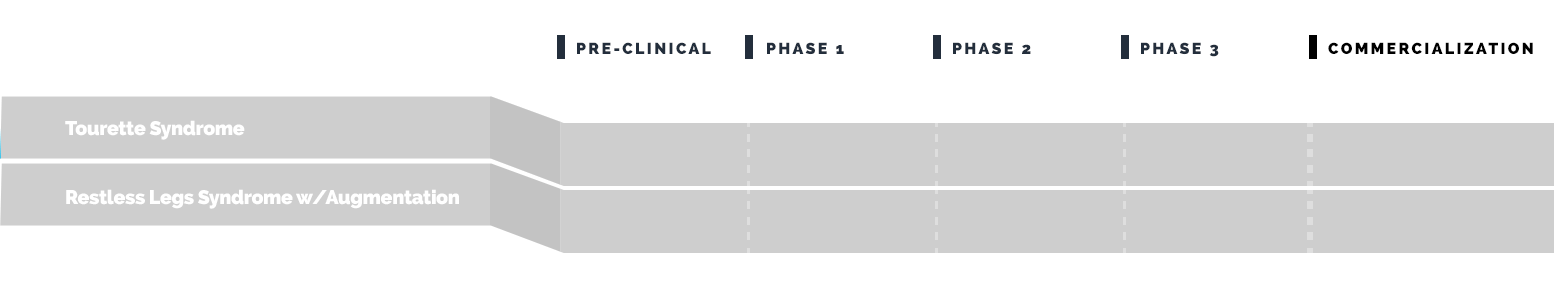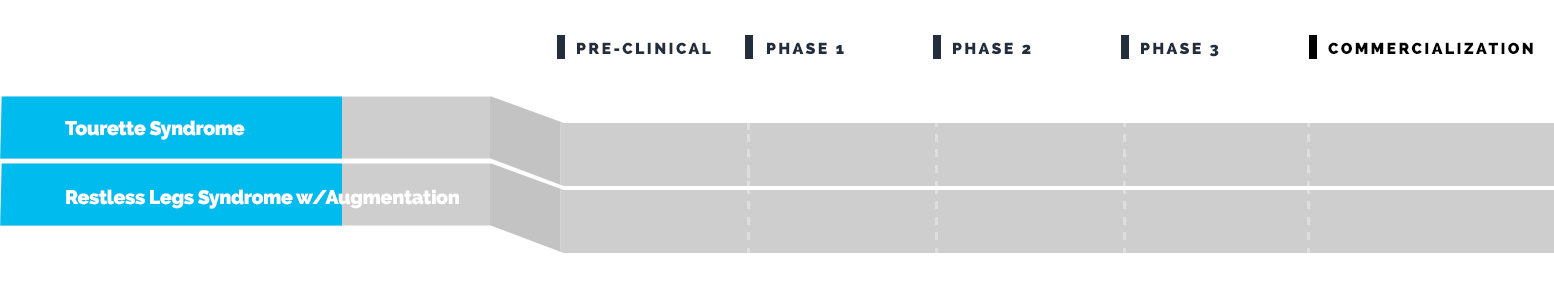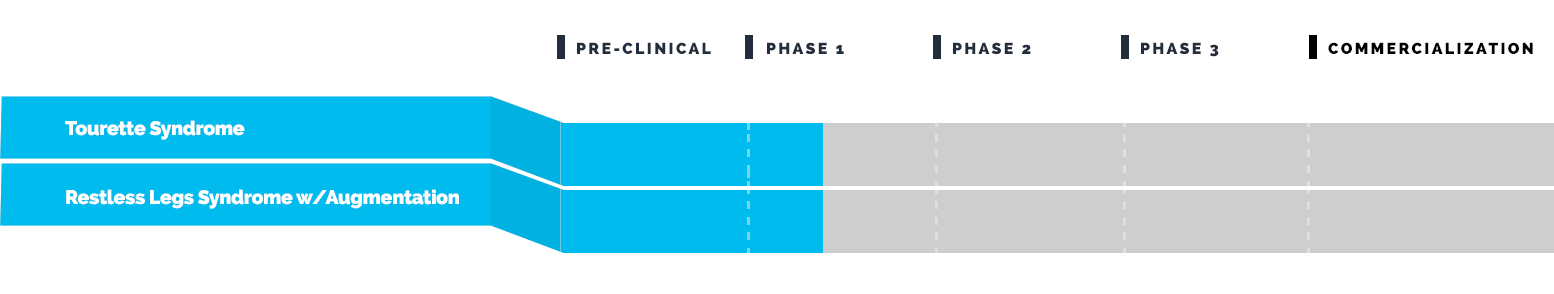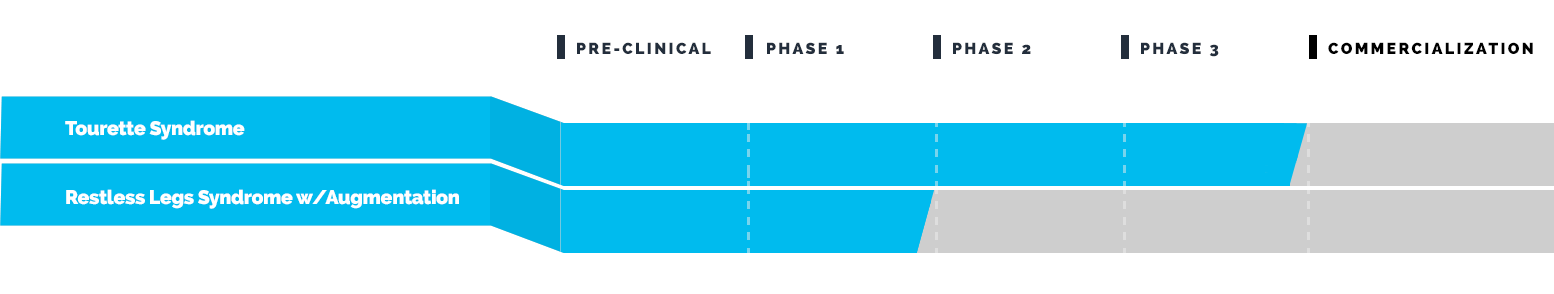
Emalex has completed its Phase 3 clinical trial to evaluate the safety and efficacy of ecopipam (EBS-101) in children, adolescents and adults with Tourette’s. This study (NCT05615220) enrolled patients in North America and Europe and was regulated by the U.S. Food and Drug Administration (FDA) and by similar regulatory agencies outside the U.S.
Further information and background on clinical trials may be found on www.clinicaltrials.gov, a service of the U.S. National Institutes of Health (NIH).
Ecopipam
Emalex’s first-in-class development candidate ecopipam, a novel investigational compound that is being studied as a potential treatment for certain central nervous system disorders, blocks the actions of the neurotransmitter dopamine at the D1 receptor. Dopamine is a neurotransmitter in the central nervous system, and its receptors have been classified into two “families” based on their genetic structure: “D1” (including subtypes D1 and D5) and “D2” (including subtypes D2, D3, and D4). D1 receptor super-sensitivity may be a mechanism for the repetitive and compulsive behaviors associated with Tourette’s. Currently approved therapies for the treatment of Tourette’s act at D2 receptors. Ecopipam has been shown to be generally well tolerated in clinical trials conducted to date and has received Orphan Drug and Fast Track designation from the FDA for the treatment of patients with Tourette’s. Adverse events affecting primarily the central nervous system have been reported in clinical trials conducted to date, including headache, fatigue, somnolence, insomnia, restlessness, anxiety, depression and rarely, suicidal ideation.
Drug Pipeline
Expanded Access Program and Policy
Emalex has completed its Phase 3 program for Tourette syndrome and has progressed to the Expanded Access Program (EAP).
This study, (EBS-101-TD-392), “An Open Labeled Expanded Access Program Intended to Provide Treatment with Ecopipam (EBS-101) to Children, Adolescents, and Adults in the U.S. with Tourette’s Disorder,” is currently enrolling.
You can contact Emalex Biosciences at medicalaffairs@emalex.com for further information about this study or click here.
Click here to view Emalex’s Expanded Access Policy.
